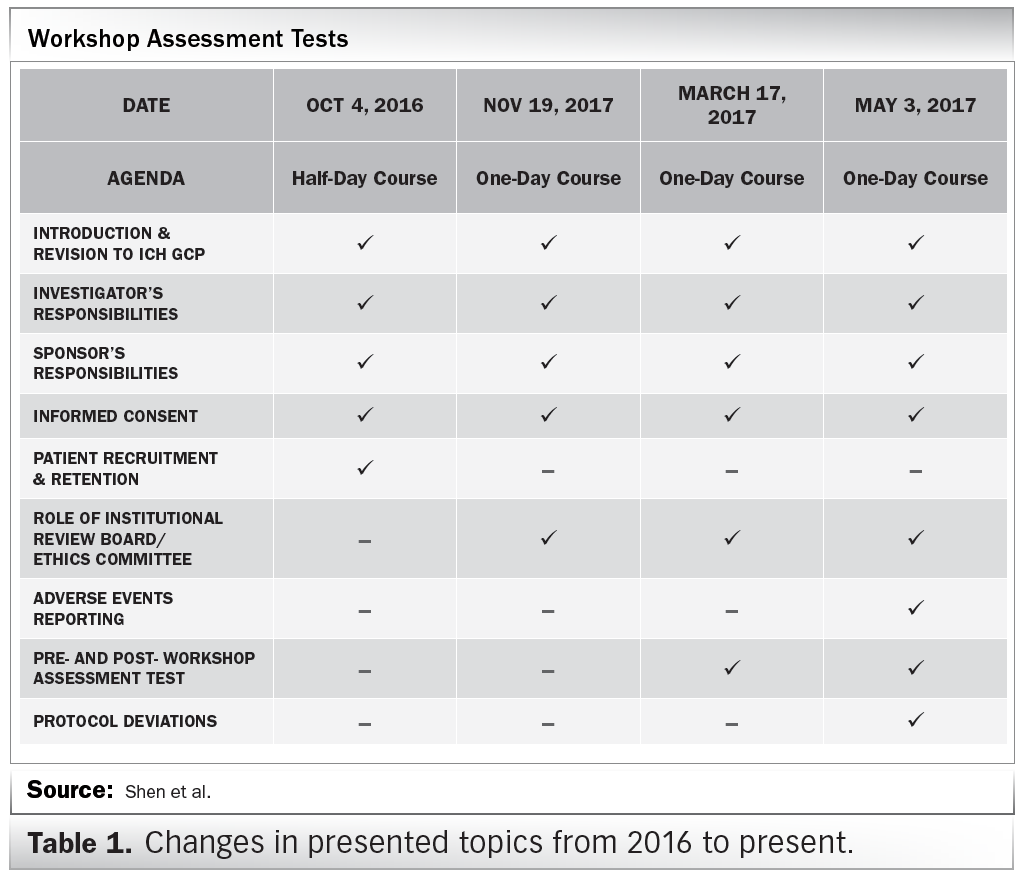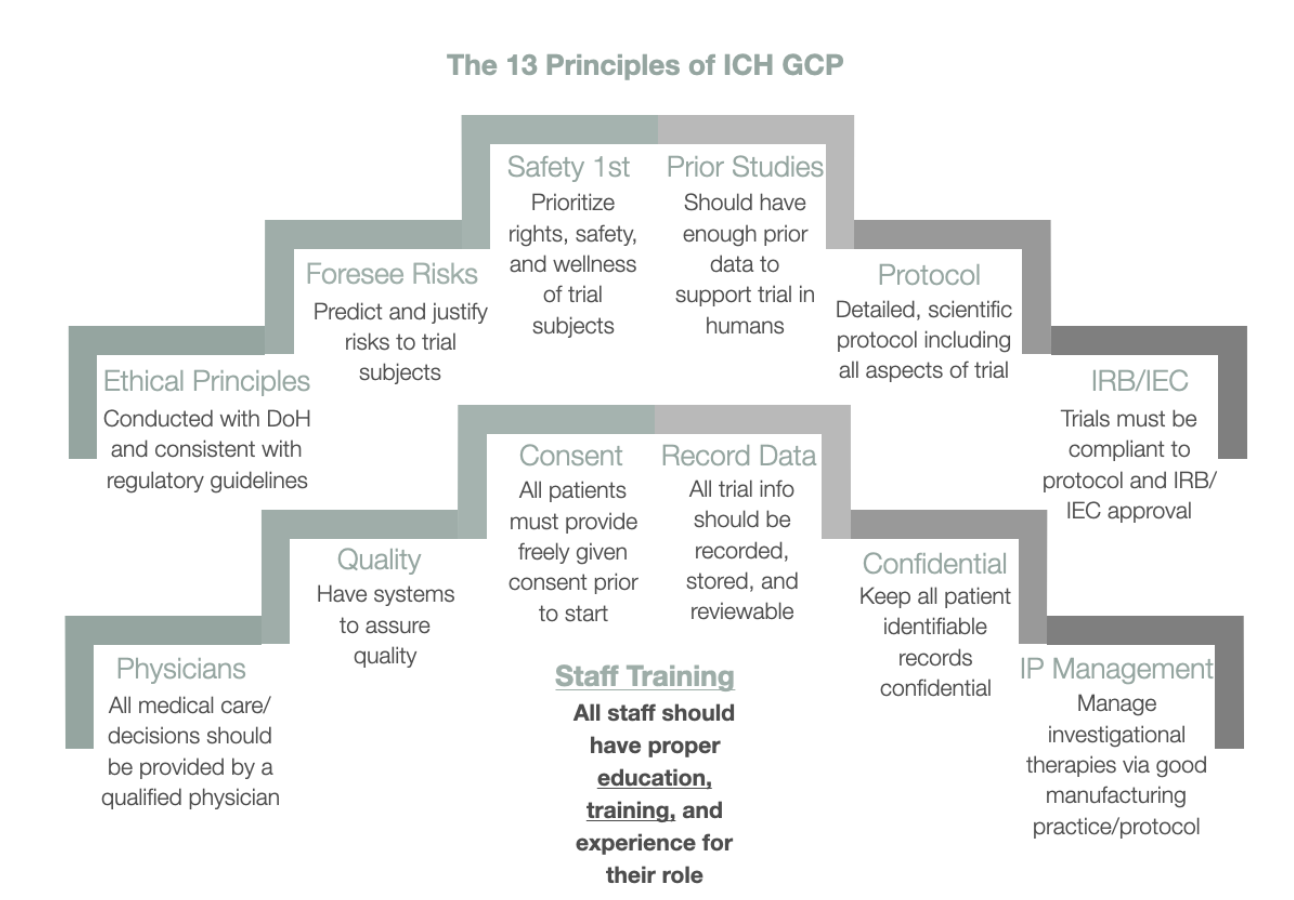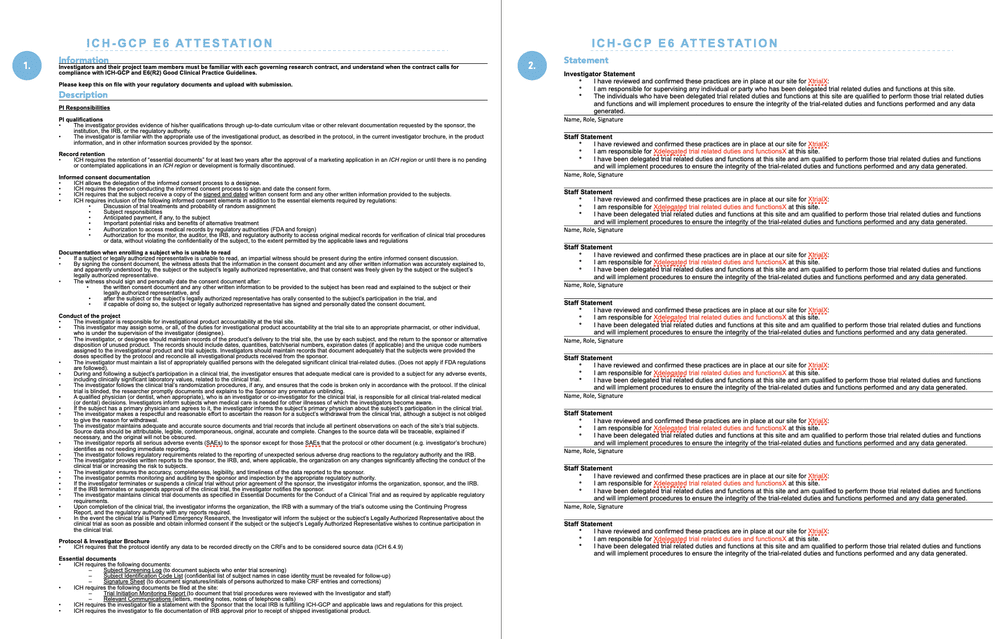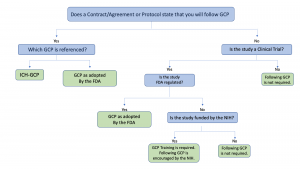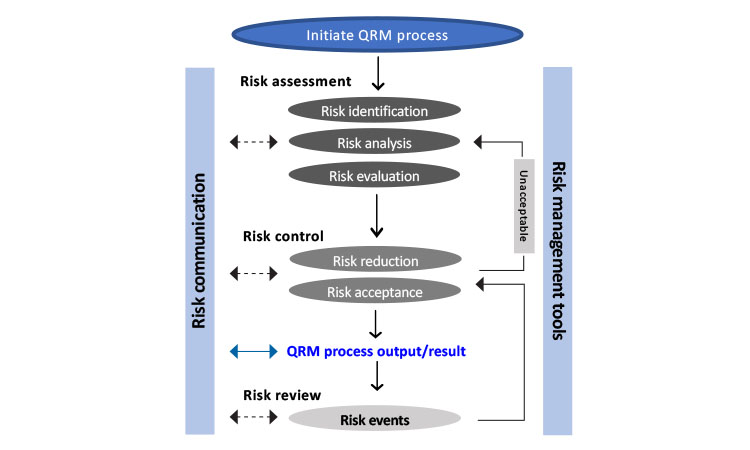
Clinical Trial Management Adaptation to ICH E6 (R2): Good Clinical Practice | Pharmaceutical Engineering

Compliance with the ICH-GCP Guidelines among the Saudi Health Care Professionals: Should Saudi Arabia Conduct Widespread ICH-GCP Training? | Semantic Scholar

The Good Clinical Practice guideline and its interpretation – perceptions of clinical trial teams in sub‐Saharan Africa - Vischer - 2016 - Tropical Medicine & International Health - Wiley Online Library